Optimal Design of Bioprinted Grafts to Promote Tissue Vascularization and Cell Viability
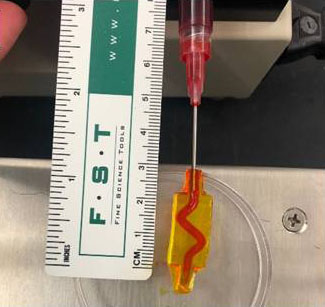
Skin is the largest organ in the human body. It functions to protect the internal organs from dangerous chemicals, extreme temperatures, and pathogens that exist in the outside world. When this organ is deeply damaged, the body cannot utilize its built-in skin regeneration mechanisms for healing. Tissue engineering of skin grafts is a promising tool to aid the body's healing processes. There are some clinical grafts currently available, but there is still a challenge with full-thickness skin grafts that are needed for patients with major burns or non-healing ulcers. A careful selection of skin graft parameters is needed to ensure the restoration of healthy tissues. An important method being explored to aid in the efficacy of artificial skin grafts is the introduction of channels that allow blood to infiltrate the graft material more quickly and provide faster transport of the necessary nutrients to the cells formed outside these channels. The ability to control the properties of these channels and bulk material has the potential to benefit the response in the body, but this requires optimization and a clear comparison between different engineering approaches that has not yet been performed in the field. For example, the diameter of these channels can change over time, altering the optimum initial channel size needed to improve the viability of the cells within the graft. Important parameters include material composition, cross-linking structure (mesh size), water content, salinity, and thickness, which can considerably alter the influence of swelling, the degradation time of the material, and the transport of blood and dissolved substances throughout the grafts.
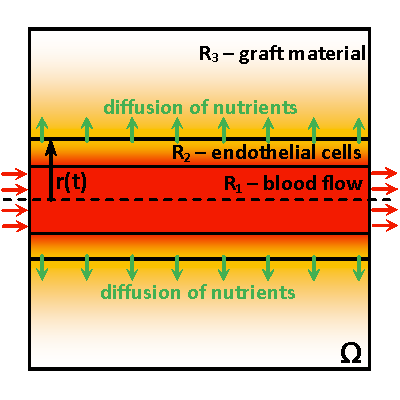
Motivated by this promising technology, this research aims to utilize modeling and optimization techniques to find the ideal parameters of the grafting material, such as cross-linking density and hydrogel material-water composition. An efficient computational framework will support mathematical and physical concepts by producing exhaustive computations for comprehensive and accurate models representing the main biophysical phenomena inside the grafts. Modern optimization algorithms will enhance this framework to enable the search for the optimal channel design. The research targets a reliable methodology to promote tissue vascularization by advising on optimal geometry of channels (diameters, mutual positioning, and shapes) and material composition and structure to ensure optimal nutrient transport throughout the graft. The computed models aim to determine the transport of nutrients and cell growth in engineered grafts with the potential to improve the healing time of patients with skin injuries and nurture more viable cells within the artificial grafts.
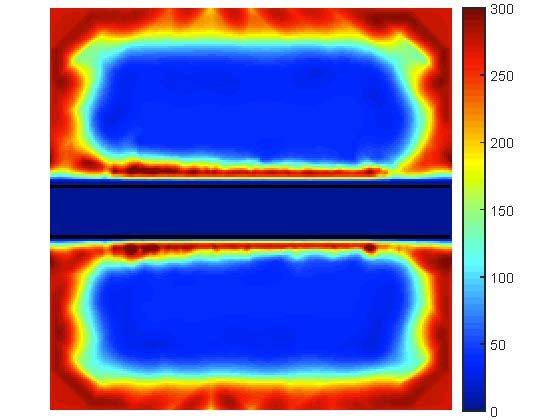
A robust computational framework created as a result of the proposed research will solve a wide range of problems: simulation of physical and biological phenomena in the graft material with the channels of a prescribed structure, reconstruction of parameters governing these phenomena, and optimal channel design. It will include computational methods for high-performance computing, solving the highly nonlinear inverse problems, and applying methods for regularization and multilevel parameterization. Every part of this framework will be carefully validated in real-based models using the in-vitro results of laboratory experiments with hydrogel materials. Once completed, it opens new horizons for successful forecasting of in-vivo models and perspectives for applying this methodology in broad medical practice. This includes grafts for a variety of tissues in addition to skin.
Project Collaborators
- Chris Bashur (Dept. of Biomedical and Chemical Engineering and Sciences, Florida Institute of Technology)
- Beste Caner (Dept. of Biomedical and Chemical Engineering and Sciences, Florida Institute of Technology)
- Nick Huynh (Dept. of Biomedical and Chemical Engineering and Sciences, Florida Institute of Technology)
- Briana Edwards (Dept. of Mathematical Sciences, Florida Institute of Technology)
References
- B. Caner, B.L. Edwards, N. Huynh, C. Bashur, and V. Bukshtynov, "Optimizing Tissue Vascularization in Bioprinted Skin Grafts", SIAM News, May 9, 2024 (the final version of the paper is available at SIAM News Blog).
Acknowledgments

Registration and travel support for presenting at the 10th International Congress on Industrial and Applied Mathematics ICIAM 2023 (Tokyo, Japan) was provided by National Science Foundation Grant DMS-2233032 and the Society for Industrial and Applied Mathematics.
